When referring to the compilation of Henry's Law Constants, please cite
this publication:
R. Sander: Compilation of Henry's law constants (version 5.0.0) for
water as solvent, Atmos. Chem. Phys., 23, 10901-12440 (2023),
doi:10.5194/acp-23-10901-2023
The publication from 2023 replaces that from 2015,
which is now obsolete. Please do not cite the old paper anymore.
|
| FORMULA: | N2F4 |
|
TRIVIAL NAME:
|
tetrafluorohydrazine
|
|
CAS RN: | 10036-47-2 |
STRUCTURE
(FROM
NIST):
|
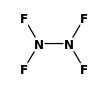
|
|
InChIKey: | GFADZIUESKAXAK-UHFFFAOYSA-N |
|
|
References |
Type |
Notes |
| [mol/(m3Pa)] |
[K] |
|
|
|
| 8.4×10−6 |
2500 |
Burkholder et al. (2019) |
L |
1)
|
| 8.4×10−6 |
2500 |
Burkholder et al. (2015) |
L |
1)
|
| 8.4×10−6 |
2500 |
Sander et al. (2011) |
L |
1)
|
| 8.4×10−6 |
2500 |
Wilhelm et al. (1977) |
L |
|
| 8.4×10−6 |
2400 |
Dean et al. (1973) |
M |
110)
|
Data
The first column contains Henry's law solubility constant
at the reference temperature of 298.15 K.
The second column contains the temperature dependence
, also at the
reference temperature.
References
-
Burkholder, J. B., Sander, S. P., Abbatt, J., Barker, J. R., Huie, R. E., Kolb, C. E., Kurylo, M. J., Orkin, V. L., Wilmouth, D. M., & Wine, P. H.: Chemical Kinetics and Photochemical Data for Use in Atmospheric Studies, Evaluation No. 18, JPL Publication 15-10, Jet Propulsion Laboratory, Pasadena, URL https://jpldataeval.jpl.nasa.gov (2015).
-
Burkholder, J. B., Sander, S. P., Abbatt, J., Barker, J. R., Cappa, C., Crounse, J. D., Dibble, T. S., Huie, R. E., Kolb, C. E., Kurylo, M. J., Orkin, V. L., Percival, C. J., Wilmouth, D. M., & Wine, P. H.: Chemical Kinetics and Photochemical Data for Use in Atmospheric Studies, Evaluation No. 19, JPL Publication 19-5, Jet Propulsion Laboratory, Pasadena, URL https://jpldataeval.jpl.nasa.gov (2019).
-
Dean, C. R. S., Finch, A., & Gardner, P. J.: The aqueous solubilities of nitrogen trifluoride and dinitrogen tetrafluoride, J. Chem. Soc. Dalton Trans., pp. 2722–2725, doi:10.1039/DT9730002722 (1973).
-
Sander, S. P., Abbatt, J., Barker, J. R., Burkholder, J. B., Friedl, R. R., Golden, D. M., Huie, R. E., Kolb, C. E., Kurylo, M. J., Moortgat, G. K., Orkin, V. L., & Wine, P. H.: Chemical Kinetics and Photochemical Data for Use in Atmospheric Studies, Evaluation No. 17, JPL Publication 10-6, Jet Propulsion Laboratory, Pasadena, URL https://jpldataeval.jpl.nasa.gov (2011).
-
Wilhelm, E., Battino, R., & Wilcock, R. J.: Low-pressure solubility of gases in liquid water, Chem. Rev., 77, 219–262, doi:10.1021/CR60306A003 (1977).
Type
Table entries are sorted according to reliability of the data, listing
the most reliable type first: L) literature review, M) measured, V)
VP/AS = vapor pressure/aqueous solubility, R) recalculation, T)
thermodynamical calculation, X) original paper not available, C)
citation, Q) QSPR, E) estimate, ?) unknown, W) wrong. See Section 3.1
of Sander (2023) for further details.
Notes
| 1) |
A detailed temperature dependence with more than one parameter is available in the original publication. Here, only the temperature dependence at 298.15 K according to the van 't Hoff equation is presented. |
| 110) |
The data from Dean et al. (1973) were fitted to the three-parameter equation: Hscp= exp( −318.29953 +15733.17858/T +44.55320 ln(T)) mol m−3 Pa−1, with T in K. |
The numbers of the notes are the same as
in Sander (2023). References cited in the notes can be
found here.
|
* * *
Search Henry's Law Database
* * *
Convert Henry's Law Constants
* * *
|

